 Image 1 of 1
Image 1 of 1


Reference Interval_EP28_CAP_COLA_CLIA_ByLCS
Here's a standalone listing description for the EP28 template:
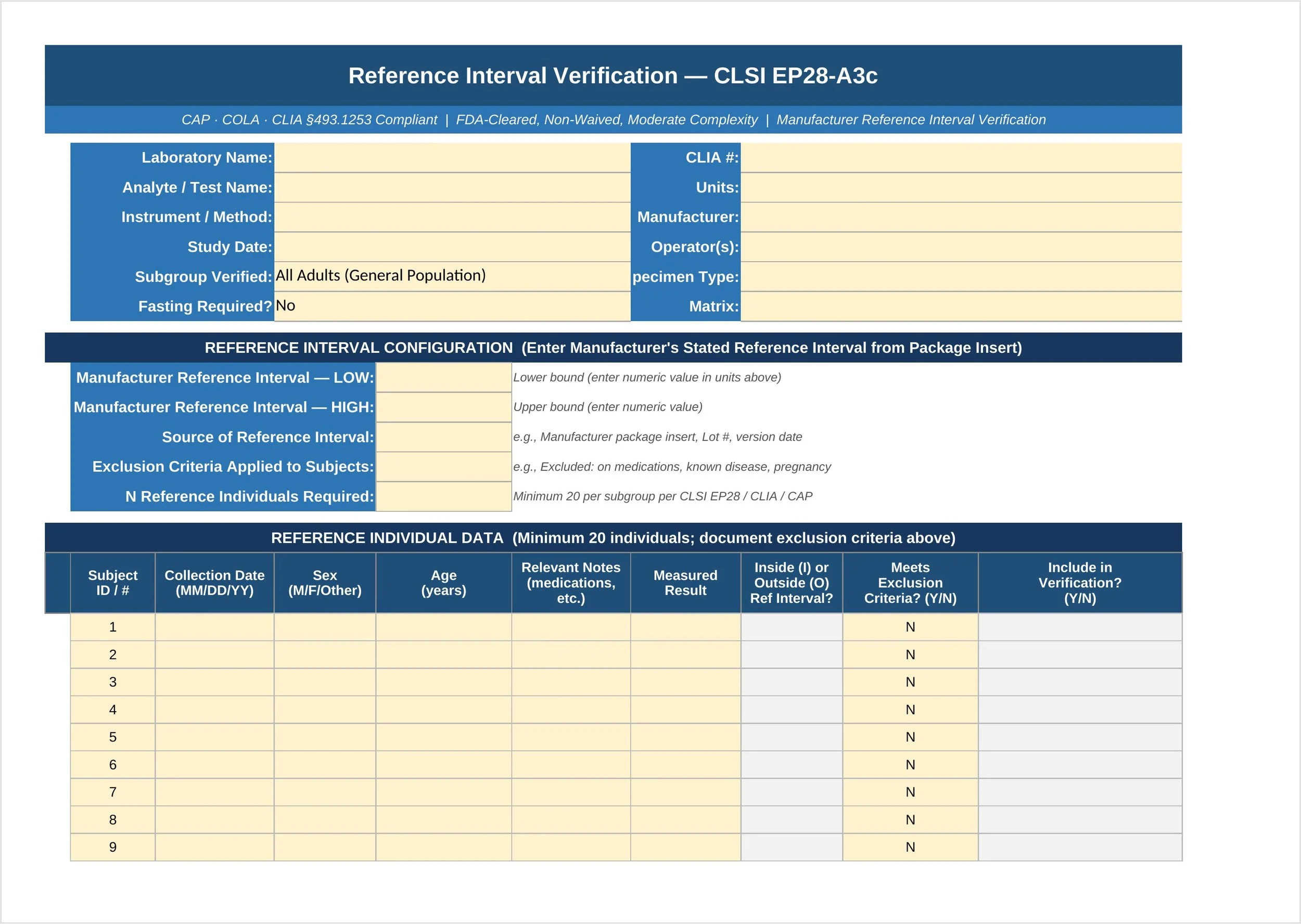
Reference Interval Verification — CLSI EP28-A3c | CAP · COLA · CLIA Compliant For FDA-Cleared, Non-Waived, Moderate Complexity Quantitative Assays
Every quantitative test your lab reports needs a verified reference interval — the range that tells clinicians what's "normal" for your patient population. CLIA (42 CFR §493.1253), CAP (GEN.41058), and COLA all require that reference intervals be verified before use. But for FDA-cleared assays, you don't need to build one from scratch. You just need to confirm the manufacturer's stated interval works for your patients.
This Excel template walks you through the CLSI EP28-A3c simplified verification method — the standard approach accepted by CAP and CLIA for FDA-cleared tests — using a minimum of 20 reference individuals.
What It Does
You run 20 specimens from healthy reference individuals, enter the results, and the template does the rest. It automatically flags each result as inside or outside the manufacturer's stated reference interval, counts the outliers, and applies the EP28-A3c 10% rule to deliver a clear, documented conclusion:
≤2 of 20 outside the interval → Reference interval is verified and acceptable to adopt
≥4 of 20 outside the interval → Interval is not verified; investigation required before patient reporting
Key Features
Supports up to 30 reference individuals with demographics and exclusion criteria tracking
Auto-calculates inside/outside status against your configured low and high limits
10% rule evaluated and documented automatically
Free-text outlier investigation field — required documentation when any results fall outside
Configurable for any subgroup (male/female, pediatric/adult, fasting/non-fasting)
Full compliance statement referencing CLIA §493.1253(b)(4), CAP GEN.41058, and COLA Principle 7
Lab director attestation and signature section
Includes detailed instructions covering: Who qualifies as a reference individual, how to apply exclusion criteria, what to do when the interval fails verification, subgroup considerations, and record retention requirements.
Provided as an unlocked Excel (.xlsx) file. Compatible with Microsoft Excel 2016 and later. Designed for verification of manufacturer-stated reference intervals for FDA-cleared, non-waived quantitative assays. Not intended for de novo reference interval establishment (which requires ≥120 subjects per CLSI EP28-A3c Section 5).
Here's a standalone listing description for the EP28 template:
Reference Interval Verification — CLSI EP28-A3c | CAP · COLA · CLIA Compliant For FDA-Cleared, Non-Waived, Moderate Complexity Quantitative Assays
Every quantitative test your lab reports needs a verified reference interval — the range that tells clinicians what's "normal" for your patient population. CLIA (42 CFR §493.1253), CAP (GEN.41058), and COLA all require that reference intervals be verified before use. But for FDA-cleared assays, you don't need to build one from scratch. You just need to confirm the manufacturer's stated interval works for your patients.
This Excel template walks you through the CLSI EP28-A3c simplified verification method — the standard approach accepted by CAP and CLIA for FDA-cleared tests — using a minimum of 20 reference individuals.
What It Does
You run 20 specimens from healthy reference individuals, enter the results, and the template does the rest. It automatically flags each result as inside or outside the manufacturer's stated reference interval, counts the outliers, and applies the EP28-A3c 10% rule to deliver a clear, documented conclusion:
≤2 of 20 outside the interval → Reference interval is verified and acceptable to adopt
≥4 of 20 outside the interval → Interval is not verified; investigation required before patient reporting
Key Features
Supports up to 30 reference individuals with demographics and exclusion criteria tracking
Auto-calculates inside/outside status against your configured low and high limits
10% rule evaluated and documented automatically
Free-text outlier investigation field — required documentation when any results fall outside
Configurable for any subgroup (male/female, pediatric/adult, fasting/non-fasting)
Full compliance statement referencing CLIA §493.1253(b)(4), CAP GEN.41058, and COLA Principle 7
Lab director attestation and signature section
Includes detailed instructions covering: Who qualifies as a reference individual, how to apply exclusion criteria, what to do when the interval fails verification, subgroup considerations, and record retention requirements.
Provided as an unlocked Excel (.xlsx) file. Compatible with Microsoft Excel 2016 and later. Designed for verification of manufacturer-stated reference intervals for FDA-cleared, non-waived quantitative assays. Not intended for de novo reference interval establishment (which requires ≥120 subjects per CLSI EP28-A3c Section 5).
